Extracellular Vesicles and Time-gated Raman Technology
Extracellular vesicles (EVs) are lipid nanocarrier released by virtually all cell types in their environment. This blog discusses EVs composition and classification, biological function and medical interest, and the use of time-gated Raman technology in EV analysis.
 Extracellular vesicles.
Extracellular vesicles.
EV metabolism related to the secretion and uptake of EVs seems to be developed early in the evolutionary stage since different findings point out the release and uptake of EVs in all of the three-life domains: Archaea, Bacteria, and Eukarya. EVs are classified in a different manner according to biogenesis pathway, cellular source, cargo, and function. Although the biological functions of EVs are not fully elucidated yet, these functions appear to vary depending on the organisms, EV type, and physiological status of the organism.
In Eukarya, particularly mammalian, EVs are particularly involved in cell-to-cell communication in both long- and short-distance within the same organism or the organism and its microbiota. EVs are also a pathway utilized by cells to discard excess metabolites, for example, lactic acid.
EVs composition and classification
EVs have heterogeneous size distributions and compositions; the biological material of the originating cells are cytosol, nucleic acid, lipids, proteins, and even entire organelles of the originating cell. Due to this heterogeneity, EVs can be classified in different ways. One of the most popular classification methods is EV classification based on the different EV biogenesis pathways; it divides EVs into three different populations known as apoptotic bodies, microvesicles, and exosomes.
Apoptotic bodies are generated during apoptosis and display a high level of phosphatidylserine on the outer surface. This EV type is quite large compared to other EV populations (>1000nm in diameter) but may also measure in the hundreds of nanometres range. Microvesicles (MVs) have sizes ranging from 50 to 1000 nm. MVs are generated and released into extracellular space by direct outward blebbing and pinching of the plasma membrane. Exosomes are among the smallest EV group and their size span from 30 to 200 nm. Exosome biosynthesis is complex and involves numerous cellular structures and organelles.
EVs contain practically all principal cell components, such as lipids, proteins, nucleic acids, metabolites, and, in some cases, organelles. The relative amount of these components differs significantly between EV subpopulations and EVs produced from different cell types.
 3D illustration of cells releasing exosomes.
3D illustration of cells releasing exosomes.
EVs biological function and medical interest
EVs are involved in cell-to-cell communication and as pathways utilized by cells to discard excess metabolites. As cell messengers, EVs exert their functions in various ways: via ligand-cell surface receptor binding, membrane fusion, and endocytosis. In physiological conditions, EVs are involved in numerous functions, such as tissue repair, wound healing, stem cell maintenance, blood coagulation, synaptic plasticity, and immune and inflammation modulation.
EVs are also involved in the pathological process and their role varies enormously between different pathology. For example, in Alzheimer’s disease, EVs carry misfolded beta-amyloids; thus they seem to be involved in the progression of the neurodegenerative disease. EVs might be also involved in cancer progression. For instance, EVs released by aggressive types of tumor cells promote similar aggressive phenotypes in other cancer cells. This includes drug resistance, increments of invasiveness, and proliferation.
Besides their physiological and pathological functions, EVs have raised interest in the scientific community as a potential diagnostic tool, and drug delivery system of a therapeutical agent. The diagnostic potential of the EVs lies in the fact that EV chemical composition is affected by the cell source, thus cancer cells derived EVs display a different molecular signature compared to EVs derived from healthy cells.
The application of EVs as a drug carrier is based on the low toxicity of EVs and the innate ability to cross tissue and cellular barriers. Additionally, evidence suggests that EVs might have innate targeting ability, thus the uptake of specific EVs is most likely done by certain cell type. EVs’ therapeutic applications are not limited to their cargo properties, EVs derived from certain cell types might be able to reduce inflammatory processes, promote tissue regeneration and promote synapsis plasticity.
Independent of the EV application or study, EV preparation should be thoroughly characterized. In this context, Raman spectroscopy can simplify routine EV analysis. The feature of the Raman spectrum depends on the chemical composition of the samples, thus the comparison of Raman spectra of different EV preparations can be used to indicate EV purity and highlight differences between EV populations.
Raman spectroscopy in the EV field is a powerful tool: it is fast, operator independent, label-free, and requires little to no sample manipulation. In addition, time-gated Raman spectroscopy provides clear spectra because it effectively suppresses the fluorescence by increasing the signal-to-noise ratio as pointed out in this publication.
Time-gated Raman assessing extracellular vesicles purity and distinguishing between different types of EVs
Here, we report a case study where we investigated the capability of Timegated® Raman in EV analysis.
We have measured the Raman spectra of erythrocyte-derived EV (EryEV) and thrombocyte-derived EV (TrEV). EryEV was purified by ultracentrifugation (UCx1) and two-time ultracentrifugation (UCx2) in which the pellet obtained from the first centrifugation was resuspended and centrifuged again, thus reducing the amount of non-EV material, mainly soluble proteins TrEV samples were purified by UCx2.
Each sample was measured 10 times, using a time-gated PicoRaman M3 spectrometer with a 532 nm pulsed laser coupled to a microprobe with a 40x objective. The measurement time was 30 seconds and the laser power was 35mW.
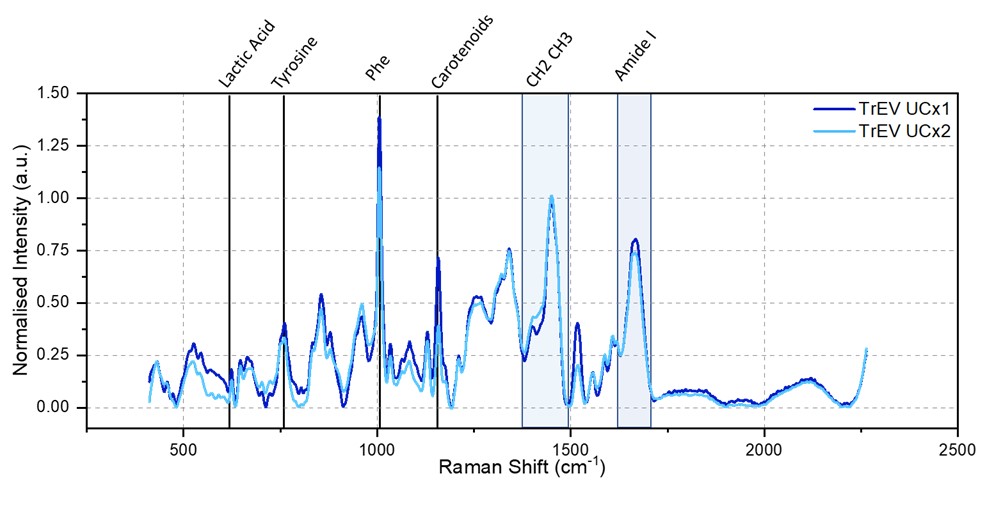 Figure 1 Timegate Raman spectra of TrEV UCx1 and TrEV UCx2. The averaged and normalized spectra of 10 measurements are reported in blue (TrEV UCx1) and light blue (TrEV UCx2) The spectral ranges of biomolecular contributions are highlighted on top of the figure.
Figure 1 Timegate Raman spectra of TrEV UCx1 and TrEV UCx2. The averaged and normalized spectra of 10 measurements are reported in blue (TrEV UCx1) and light blue (TrEV UCx2) The spectral ranges of biomolecular contributions are highlighted on top of the figure.
Raman spectra of TrEV UCx1 and TrEV UCx2 (figure 1) show similar features; Raman peak associated with lactic acids, tyrosine, phenylalanine, carotenoids, CH2 CH3 stretching, and Amide I are present in both EV preparation. However, the relative intensity is different; TrEV UCx2 displays a reduction in the intensity of protein-associated peaks (tyrosine, phenylalanine, and Amide I) indicating a lower number of proteins. Since aggregate and soluble proteins are one of the main impurities in these samples, we can confirm that UCx2 samples have a higher degree of purity.
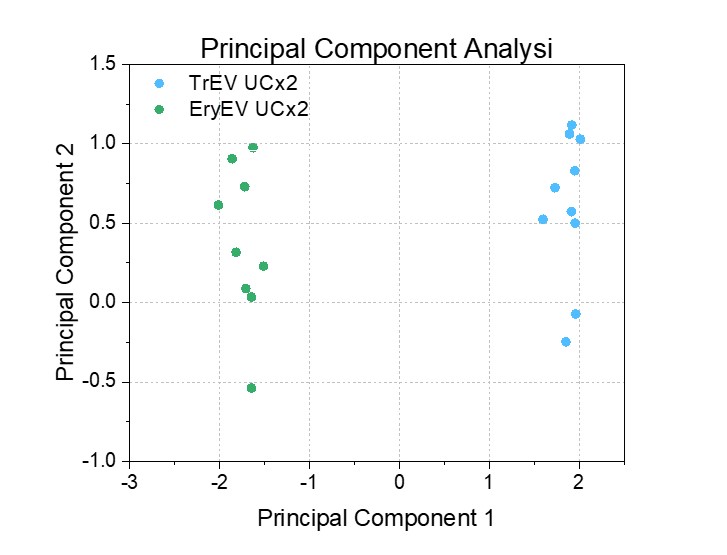 Figure 2 Principal component analysis result.
Figure 2 Principal component analysis result.
Subsequently, we compared the spectra of two EV populations (Figure 2). Principal component analysis (PCA) indicates clear differences between TrEV and EryEV samples.
Conclusion
Time-gated Raman technology is proven to be a powerful technique to assess EV purity (UCx1 vs UCx2 samples) and is able to distinguish between different types of EVs.
Read also our last blog Studies of Timegated® Raman in Life Sciences in which we introduced a peer-reviewed article about Timegated® Raman monitoring the discrimination of extracellular vesicles.
Author
 This blog was written by Timegate Instruments’ Senior Application Specialist Dr. Jacopo Zini. Jacopo has a Ph.D. in Pharmacy. His work is focused on the biopharmaceutical application of Raman spectroscopy. In his studies, he applied various photonics techniques in the characterization of complex biological samples. Read more about him and the whole Timegate team.
This blog was written by Timegate Instruments’ Senior Application Specialist Dr. Jacopo Zini. Jacopo has a Ph.D. in Pharmacy. His work is focused on the biopharmaceutical application of Raman spectroscopy. In his studies, he applied various photonics techniques in the characterization of complex biological samples. Read more about him and the whole Timegate team.